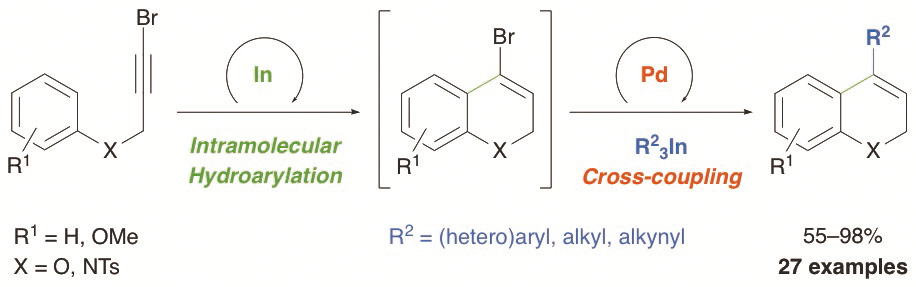
A sequential one-pot indium-catalyzed intramolecular hydroarylation (IMHA) of bromopropargyl aryl ethers and amines, and palladium-catalyzed cross-coupling reaction using triorganoindium reagents (R3In) has been developed. In this transformation, the IMHA of 3-bromo-2-propynyl aryl ethers under indium(III) catalysis, proceeds regioselectively through a 6-endo-dig pathway to afford 4-bromo-2H-chromenes. Subsequent palladium-catalyzed cross-coupling with R3In gives 4-substituted-2H-chromenes in one-pot. This sequential transformation was extended to 3-bromo-2-propynyl-N-tosylanilines to afford 4-substituted-1,2-dihydroquinolines. The dual-catalyzed procedure takes place efficiently with a variety of propargyl aryl ethers and amines and R3In (R = aryl, heteroaryl, alkyl or alkynyl), showing the efficiency of these organometallics and proving the compatibility of indium and palladium in catalysis.